- 30 januari 2018
- | 3 min. leestijd
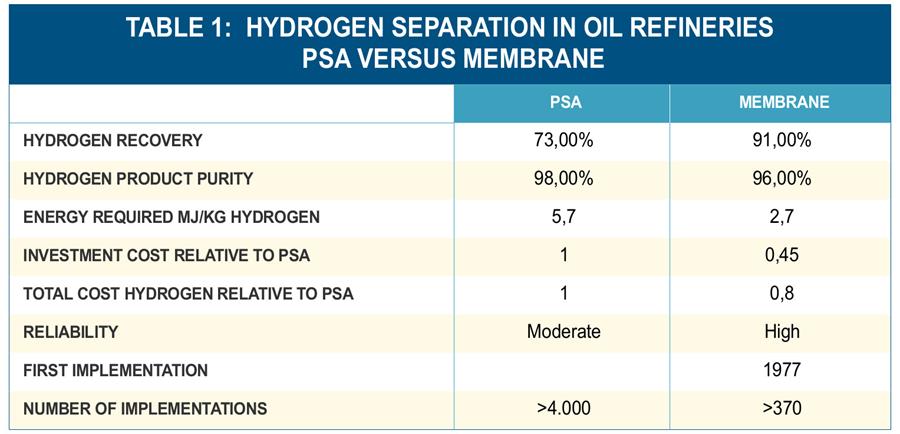
Hydrogen Membrane Separation
Several Hydrogen Membrane Separation techniques are used in oil refineries to produce hydrogen from hydrogen production units such as naphtha reforming or natural gas steam reforming. How do these compare?
Sorry, dit is een premium artikel
Dit artikel is exclusief beschikbaar voor abonnees. Abonneer je nu en krijg toegang tot alle artikels.
Ontdek onze abonnementsformules